
From these data, it can be determined whether the reaction in Eq 1 is endothermic or exothermic. (The ion reacts with free to form solid AgCl and thus removes from the solution.) Part 3: The dependence of the Equilibrium constant on temperature is determined. Part 2: Free Cl is removed from the final solution from Part 1 adding controlled amounts of AgNO3 solution. After each stepwise addition of HCl, the simulation provides the concentrations of the reactants and products from which you can determine the equilibrium constant, K for the reaction shown in (Eq 1). There are three parts to the simulation: Part 1: Concentrated HCl is added stepwise to a given volume of of known concentration. This simulation permits you to observe the colors and gives the concentrations of all species as reagents are added. When excess ion is added to a solution of the blue complex forms and the following equilibrium is established: (aq) 4 (aq) (aq) 6 H2O (l) (Eq 1) We can use the different colors to indicate the relative concentrations of the two complexes at equilibrium. 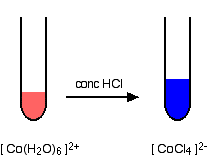
Cobalt (II) ion exists in aqueous solution as the complex ion where 6 water molecules, acting as Lewis bases, make coordinate bonds to Co donating pairs of electrons into vacant orbitals of the This results in a red solution. It is recommended that you review the chapter about equilibrium in your lecture text. Preview text CHEMICAL EQUILIBRIUM Le PRINCIPLE Virtual Lab Step Step Instructions This virtual lab should help your understanding of Le Principle observing the effect of induced stress on the equilibrium distribution of two cobalt(II) complexes in aqueous solution. Exp 2 Kinetic Blues Reaction Order and Rate Laws.The Organic Synthesis of Aspirin from Salicylic Acid in a Non-Aqueous Medium.So, as you can see, all these changes are undoubtedly in accordance with Le Chatelier's Principle. If you add #"AgNO"_3#, which will remove #"Cl"^(-)# ions from the solution, the equilibrium will shift to the left and the color will be pink again. If you add #"HCl"#, which will increase the concentration of #"Cl"^(-)"# ions, the equilibrium will shift to the right and the color will be blue. If you add water, the equilibrium will shift to the left and the color will be pink. Likewise, any other stress applied to the equilibrium will produce a color change in the solution. When the solution is being cooled, heat is being removed from the equilibrium, which means that a shift towards to the reactants will take place - the solution will turn pink, the color of the #^(2+)# ion. This shift in the equilibrium will turn the solution blue, the color of the #CoCl_4^(2-)# ion. the solution is being heated, the equilibrium will shift in the direction of the products. We can say that heat is a reactant in this equilibrium. The forward reaction is endothermic, which means the equilibrium could be written as The #^(2+)# ion is pink, while the #CoCl_4^(2-)# ion is blue. 
In aqueous solution, the following equilibrium will be established between the following two ions: When dissolved in water, both #"CoCl"_2#, and #"CoCl"_2*6"H"_2"O"# will produce the complex ion #^(2+)#. 
The main species involved in the reaction is cobalt (II) chloride, or #"CoCl"_2#, which usually comes in the form of a hexahydrate, #"CoCl"_2*6"H"_2"O"#. Yes, the color changes that accompany the heating or cooling of the equilibrium mixture are very much in accordance with Le Chatelier's Principle.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
